Research interests
Electron transfer
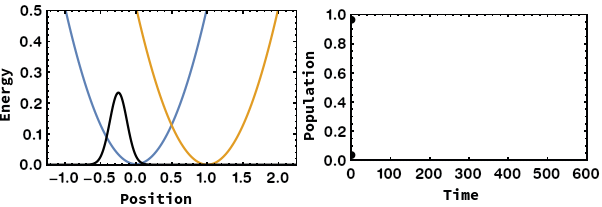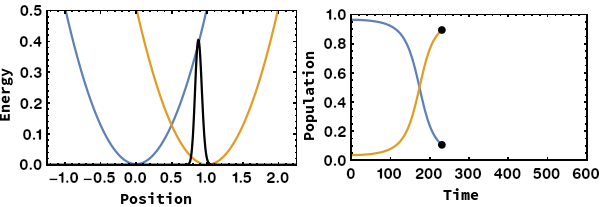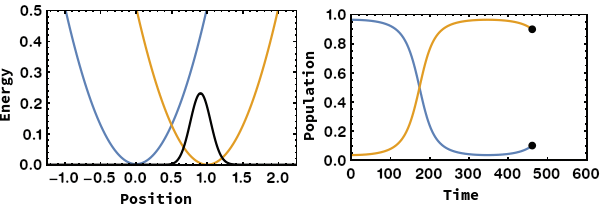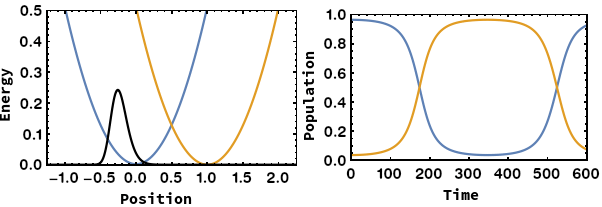
Being able to control electron transfer in mixed-valent systems is a promising way towards molecular electronics (wires or switches). We focus on unravelling the mechanisms of electron transfer using quantum chemical calculations. We were able to identify the nuclear coordinate of the Marcus–Hush model, which is the motion that drives the electron transfer. By application of this methodology we showed that: i) Electron transfer can proceed via heavy-atom tunnelling in small organic radiacal anions. ii) additional symmetric dimensions of the Marcus–Hush model that alters the electronic coupling between the redox centers is necessary for bridged systems like Creutz–Taube ion. iii) The famous square scheme of proton-coupled electron transfer can be transfered from the energy doiman into the real space by using the electron transfer coordinate.

- A. Šrut, B. J. Lear, V. Krewald, The Marcus dimension: identifying the nuclear coordinate for electron transfer from ab initio calculations, Chem. Sci., 2023, 14, 9213–9225.
- A. Šrut, B. J. Lear, V. Krewald, Symmetric Electron Transfer Coordinates are Intrinsic to Bridged Systems: An ab Initio Treatment of the Creutz–Taube Ion, Angew. Chem. Int. Ed., 2024, 63, e202404727.
- A. Šrut, M. Diefenbach, M. L. Kronenberger, B. J. Lear, V. Krewald, Mapping proton-coupled electron transfer with real space dimensions, ChemRxiv, 2025, DOI: 10.26434/chemrxiv-2025-36d35.
Biological systems
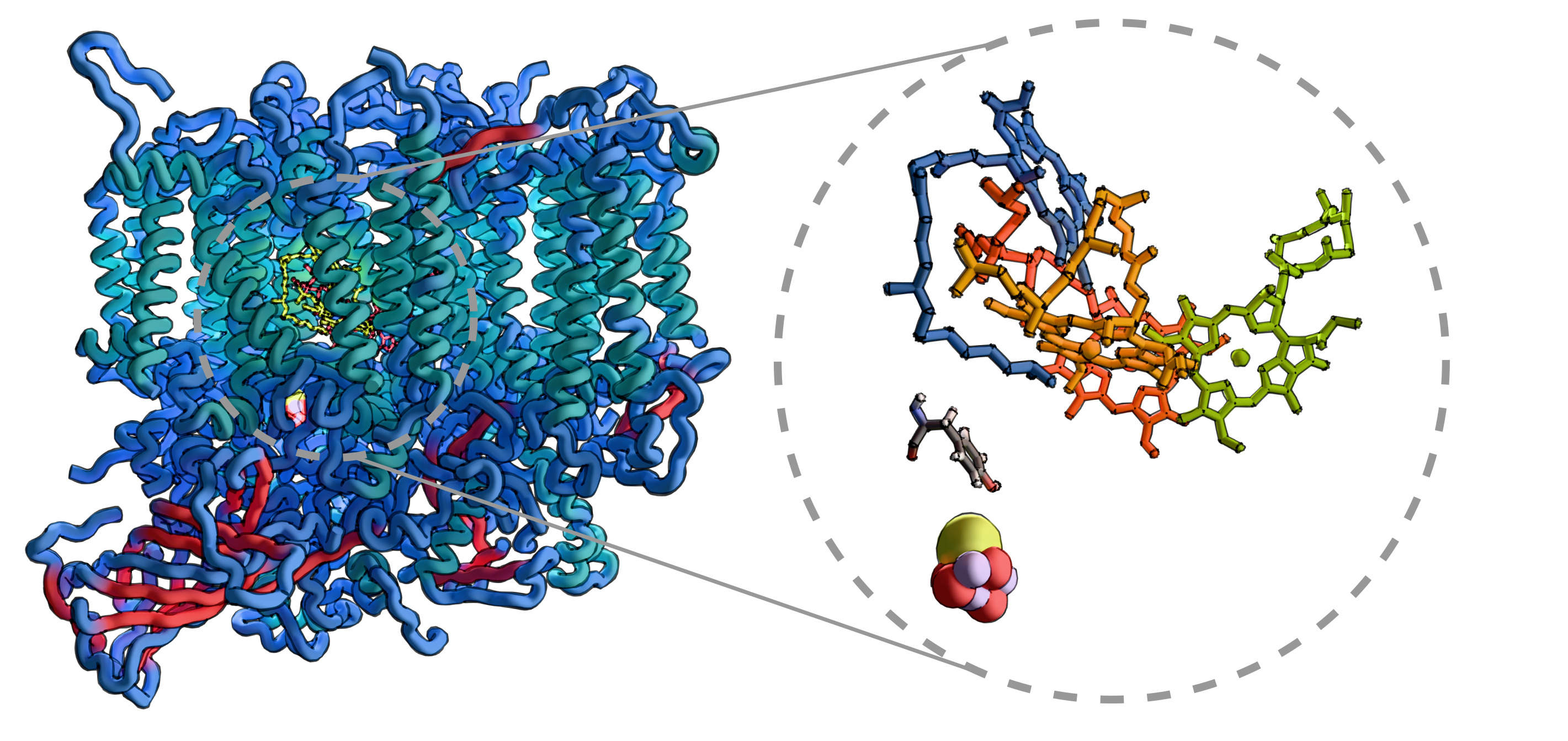
Nature's design of electron and energy transfer pathways are often fascinating. Some of these processes are reported to occur on a ultrafast time scales or with very high quantum efficiencies. The kinetics of these processes should be studied quantum mechanically also taking into account the interaction with environment. This can be realized for example with open quantum system simulation. One, however, must start with reasonable parameters for the system Hamiltonian.
Nuclear quantum dynamics
Nuclear quantum effects can play a vital role in correct interpretation of spectroscopic measurement or reaction kinetics. We use ab initio data to parameterize low-dimensional models. One can then employ grid based wave packet dynamics to calculate spectroscopic properties or reaction rates. Check out the GitHub page of QD-Engine code.